Are you looking for an answer to the topic “which of the following has the lowest entropy apex“? We answer all your questions at the website barkmanoil.com in category: Newly updated financial and investment news for you. You will find the answer right below.
Hence, oxygen gas at 0K temperature will have the lowest entropy.entropy. A thermodynamic measure of the disorder in a system. entropy change. The difference in entropy between the final and the initial states of a system, given by the equation ΔS = Sfinal – Sinitial.What most determines the entropy of a solid? The crystalline structure of the solid.

What is entropy apex?
entropy. A thermodynamic measure of the disorder in a system. entropy change. The difference in entropy between the final and the initial states of a system, given by the equation ΔS = Sfinal – Sinitial.
What most determines the entropy of a solid apex?
What most determines the entropy of a solid? The crystalline structure of the solid.
What is entropy? – Jeff Phillips
Images related to the topicWhat is entropy? – Jeff Phillips

How does a change in phase affect entropy changes in a reaction?
When a phase change occurs – that is, a substance moves from one region of its phase diagram to another – the entropy of that substance changes even as its temperature stays the same. A substance in solid phase has low entropy; in liquid phase, it has medium entropy; in gas phase, it has high entropy.
Which state of matter has the highest entropy?
Explanation: Entropy by definition is the degree of randomness in a system. If we look at the three states of matter: Solid, Liquid and Gas, we can see that the gas particles move freely and therefore, the degree of randomness is the highest.
What is entropy Brainly apex?
Entropy is the degradation of the matter and energy in the universe to an ultimate state of inert uniformity. kaypeeoh72z and 70 more users found this answer helpful. heart outlined. Thanks 67.
What does S 0 mean apex?
what does S > 0 mean? the system has become more random.
What elements have the highest entropy?
Therefore hydrogen has the highest Entropy.
See some more details on the topic which of the following has the lowest entropy apex here:
Which phase has the lowest entropy? – Answers
The lowest entropy is for ice. What is entropy and which phase of matter has the the highest entropy? Plasma · Is …
Which of the following has the lowest entropy a liquid class 11 …
Which of the following has the lowest entropy: a liquid, a gas, or a crystalline solid? Answer. Verified. 72.6k+ views.
B Which phase has the lowest entropy Why How does a …
How do the following factors affect the spontaneity of a reaction? (2 points)i. Enthalpy (1 point)Enthalpy affects the spontaneity of a reaction because if heat …
You Asked: Which Physical State Has The Lowest Entropy?
3 Which phase has the highest entropy apex? … Answer: The solid phase has the lowest entropy. Additionally, which of the following is the lowest entropy?
In which process is entropy decreased?
When heat is removed, the entropy decreases, when heat is added the entropy increases.
Which represents a decrease in entropy?
Explanation: Entropy decreases when the number of moles of gas decreases during a reaction. In the case of the correct answer, the number of moles of gas decreases from two to one. When a substance goes from a solid to a gas (sublimation) or from a liquid to a gas (evaporation), entropy increases.
What phase has the lowest entropy?
Solids have the fewest microstates and thus the lowest entropy.
What would cause entropy to decrease in a reaction apex?
Answer: The products becoming more ordered than the reactants.
Why is change in entropy lower for higher temperatures?
Entropy increases as temperature increases. An increase in temperature means that the particles of the substance have greater kinetic energy. The faster moving particles have more disorder than particles that are moving more slowly at a lower temperature.
Which has least entropy?
Hence, oxygen gas at 0K temperature will have the lowest entropy. The third law of thermodynamics states that the entropy approaches zero at absolute zero for perfectly crystalline substances.
Which condition has lowest entropy?
Answer and Explanation: The answer is (a) Solid. The entropy of substances with different states are different since the arrangement of the molecules are different.
15.2 Entropy and spontaneity (HL)
Images related to the topic15.2 Entropy and spontaneity (HL)
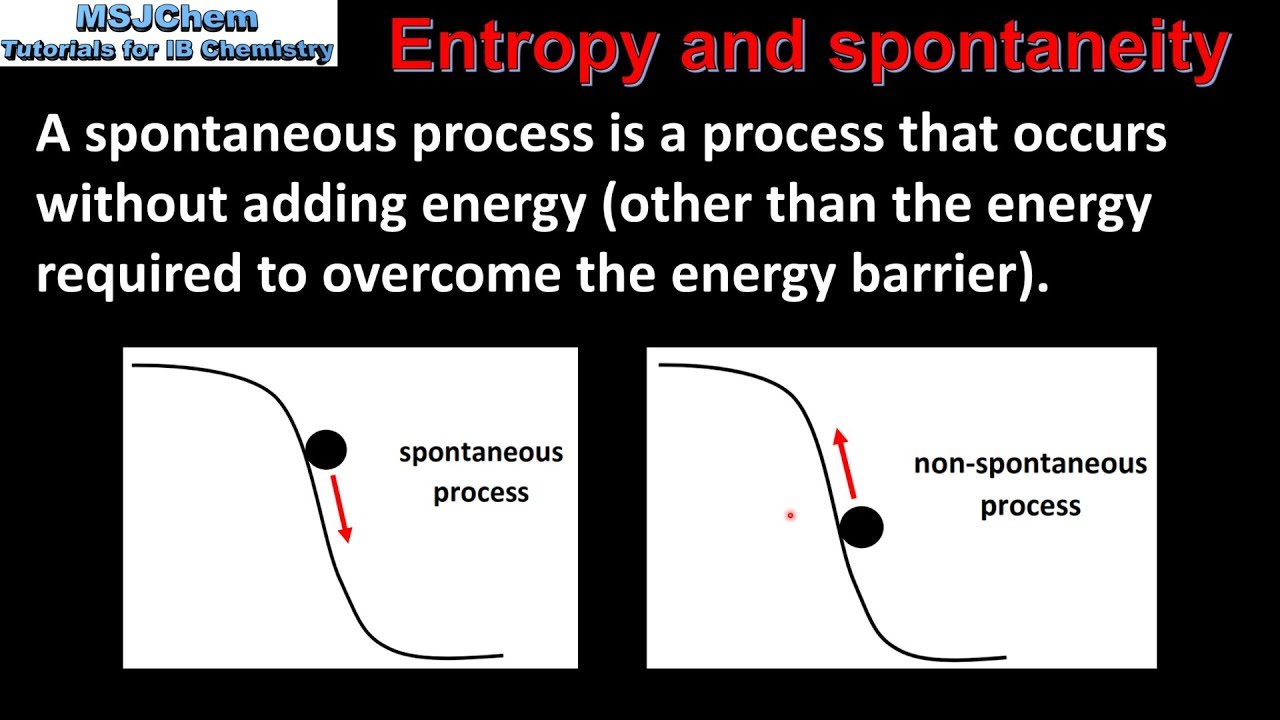
Are gases high or low entropy?
Gases have higher entropies than solids or liquids because of their disordered movement. That means that during a reaction in which there is a change in the number of molecules of gas present, entropy will change.
Which of the following best defines entropy?
Entropy is defined as the measurement of degree of randomness or in other words, it is the increase in the disorganization within a system.
What does the second law of thermodynamics say about entropy apex?
The second law of thermodynamics states that the total entropy of a system either increases or remains constant in any spontaneous process; it never decreases.
What characteristic best describes the low entropy associated with solids?
What characteristic best describes the low entropy associated with solids? The atoms are arranged in a rigid structure.
What does S 0 mean in physics?
s0 = initial position (the position at the beginning of some event) s = final position (the position at the end of some event)
What does Delta S 0 mean?
Negative delta S (ΔS<0) is a decrease in entropy in regard to the system. For physical processes the entropy of the universe still goes up but within the confines of the system being studied entropy decreases. One example is a freezer with a cup of liquid water in it.
What does S mean in a reaction?
(s) indicates that the substance is in a solid state. an alternative way of representing a substance in a solid state. (aq) indicates that the substance is dissolved in water – the aq comes from aqueous.
Which state of matter has the lowest entropy?
Because the particles of solids are found in an organized manner with little movement, it can be said that solids have the lowest entropy out of the three states of matter.
Which of the following has the most entropy quizlet?
A gas has the highest entropy, whilst a solid has the least.
How do you know which has greater entropy?
The entropy of a substance increases with its molecular weight and complexity and with temperature. The entropy also increases as the pressure or concentration becomes smaller. Entropies of gases are much larger than those of condensed phases.
What is a simple definition of entropy?
entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.
What is entropy in simple terms?
The entropy of an object is a measure of the amount of energy which is unavailable to do work. Entropy is also a measure of the number of possible arrangements the atoms in a system can have. In this sense, entropy is a measure of uncertainty or randomness.
How To Get Into Bot Lobbies In Apex Legends Season 13
Images related to the topicHow To Get Into Bot Lobbies In Apex Legends Season 13

What is entropy Mcq?
Entropy is a measure of randomness or disorder in the system. Entropy is a thermodynamic function and is denoted by S. The higher the entropy more the disorder in the isolated system. A change in entropy in a chemical reaction is related to the rearrangement of atoms from reactants to products.
What is entropy quizlet?
Entropy. It is the measure of disorder (randomness) in a system. The thermochemical variable ‘S’ stands for the amount of randomness in a system. Phases of matter involving entropy.
Related searches to which of the following has the lowest entropy apex
- what does entropy measure
- which of the following describes the entropy change as a solution is made from a liquid and solid
- what does a negative change in entropy indicate
- which phase has the lowest entropy apex
- which phase has the highest entropy apex
- what happens to the entropy when a solution is made
- which of the following has the highest entropy apex
- what does entropy measure apex
- what is the entropy of a solid based on apex
- what is enthalpy apex
- which element has highest entropy
- what happens to the entropy in the reaction i2s i2g
- what does the second law of thermodynamics say about entropy apex
- what does the second law of thermodynamics say about entropy
- what happens to the entropy in the reaction i2(s) i2(g)
- what is entropy apex
- which of the following would cause entropy to decrease
Information related to the topic which of the following has the lowest entropy apex
Here are the search results of the thread which of the following has the lowest entropy apex from Bing. You can read more if you want.
You have just come across an article on the topic which of the following has the lowest entropy apex. If you found this article useful, please share it. Thank you very much.