Are you looking for an answer to the topic “what is the mass of exactly 500 sulfur atoms“? We answer all your questions at the website barkmanoil.com in category: Newly updated financial and investment news for you. You will find the answer right below.
Keep Reading
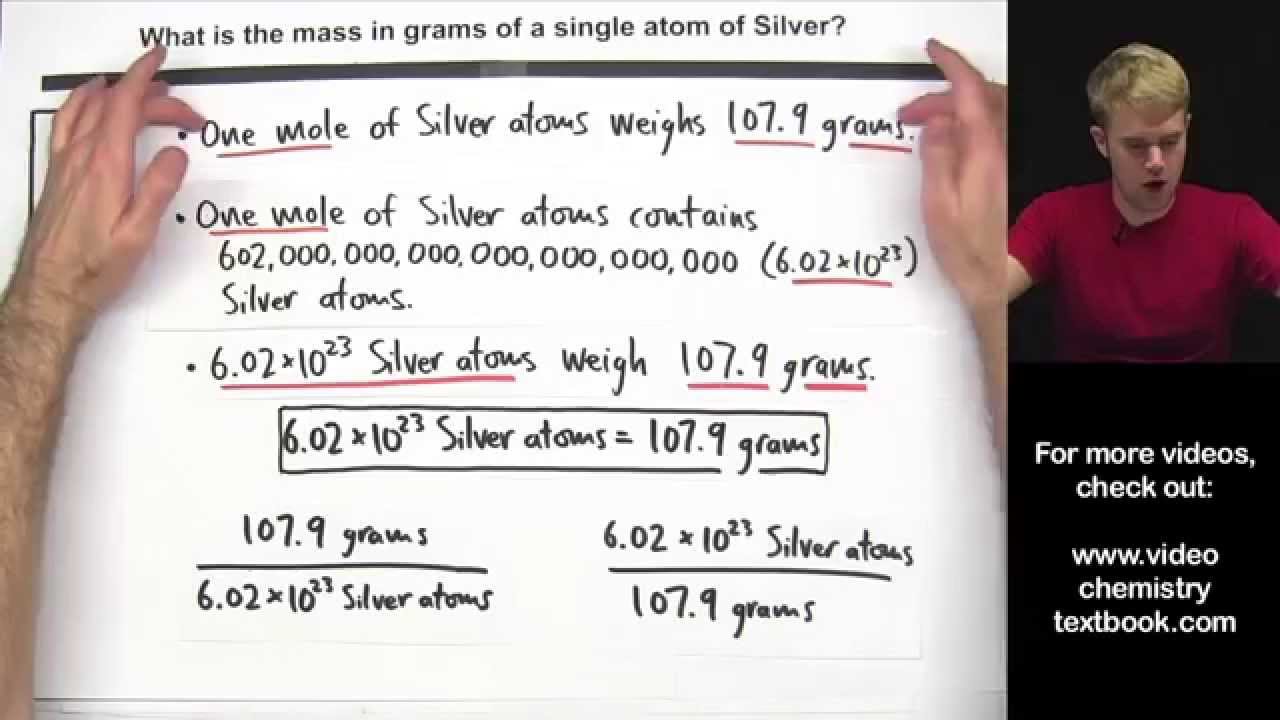
What is the mass of 1 atom of sulfur?
One atom of sulfur has a mass of 32.07 amu; one mole of S atoms has a mass of 32.07 g. For compounds, the molecular mass (in amu) is numerically the same as the mass of one mole of the compound in grams. Skill 3-1 Calculate the molecular mass of a compound as the sum of the atomic masses of its elements.
What is the mass of 500 atoms of iron?
So the 500 iron atoms weigh 4.63⋅10−20grams .
Calculate the Mass of a Single Atom or Molecule
Images related to the topicCalculate the Mass of a Single Atom or Molecule
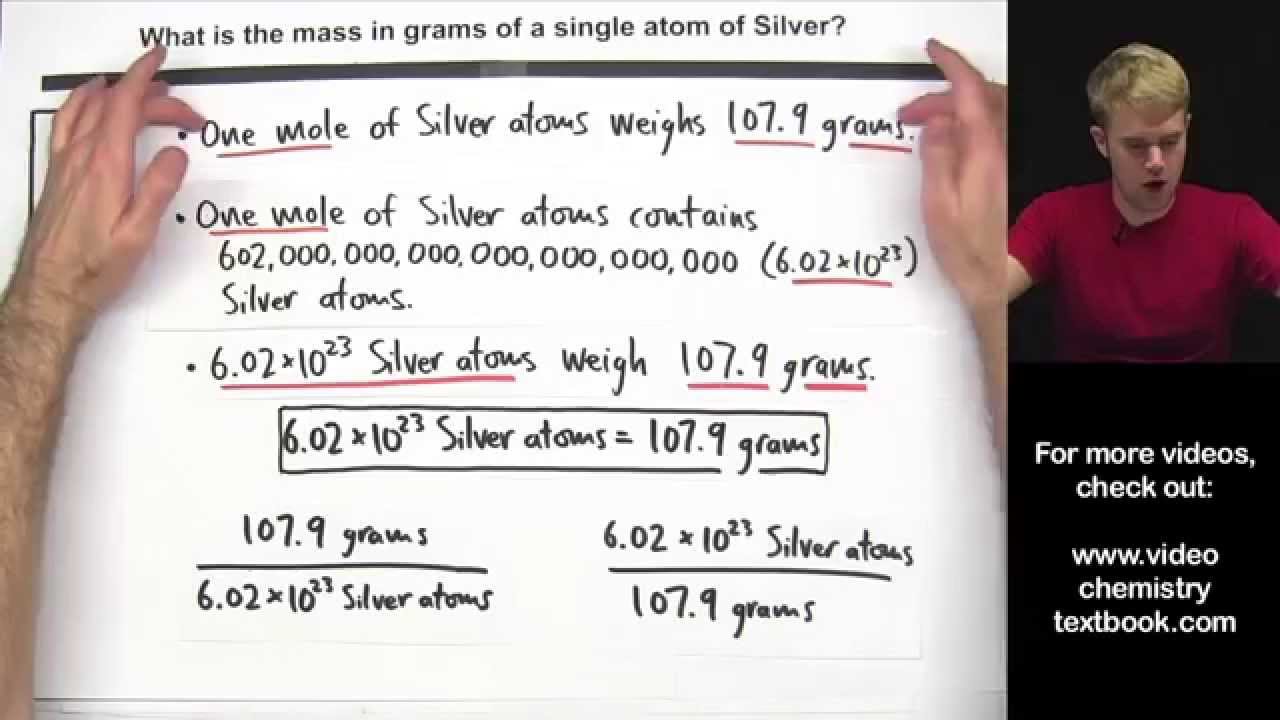
How do you find the mass of a sulfur atom?
Molar mass of sulphur = 8 X 32 = 256 g.
What is the mass of 6.022 x10 23 sulphur atoms?
We know that NA,Avogadro’s number, 6.022×1023 of sulfur atoms have a mass of 32.06⋅g precisely.
How many atoms are in 1g of sulfur?
6.022 × 1023 (Avogadro’s number) atoms are present in one gram atomic mass of a substance.
How do u calculate atomic mass?
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
What is the mass of a sulfur 32 atom?
sulfur-32 atom (CHEBI:37979) The stable isotope of sulfur with relative atomic mass 31.972071.
See some more details on the topic what is the mass of exactly 500 sulfur atoms here:
What is the mass of exactly 500 sulfur atoms? – ECHEMI
Result of one single atom of sulphur = ~ 5.3245184×10^-23 grams. Result of 500 atoms of sulphur = ~ 2.6622592×10^-20 grams.
what is the mass of exactly 500 sulfur atoms? – AnswerPrime …
5.325 e-23 * 500 = 2.663 e-20 g. Answer 2. 32g of sulphur is one mole. One mole of any substance contains 6.022045 x 10^23 mol^-1 atoms …
what is the mass of exactly 500 sulfur atoms? – ForNoob
5.325 e-23 * 500 = 2.663 e-20 g. 32g of sulphur is one mole. One mole of any substance contains 6.022045 x 10^23 mol^-1 atoms – the Avogadro …
Best Answer: What Is The Mass Of Exactly 500 Sulfur Atoms? – Ask-rk
3 How do you convert atoms to mass? 4 What is the mass in grams of one 1h atom? 5 How is mol calculated? 6 What is the mass of one mole of Sulphur atoms Class 9 …
How do you calculate sulphur?
Liters of water intake is multiplied by mg S/L = intake of sulfur in mg. Divide by 1000 for intake of sulfur in grams. Daily dry matter intake = 2-3% body weight in kg. %SH2O on DMI basis = total intake of S from water in grams divided by kg DMI divided by 10.
What is the atomic mass of S8?
Molar mass of Sulphur (S8) = 8 x Molar mass of S = 8 x 32 = 256 g.
What is the mass of 3.09 * x10 24 atoms of sulfur in grams?
Similarly, 3.09 times 10 ways to part 24 atoms of sulfur will weigh 32 divided by 6.22 times 10 days to part 23 multiplied by 3.9 times 10. Raise to part 24. From this, the mask of sulfur atom would comes out to be 164.2 g and 164.2 g is required answer.
Measuring Atomic Mass | Atoms and Molecules | Don’t Memorise
Images related to the topicMeasuring Atomic Mass | Atoms and Molecules | Don’t Memorise

What is the mass of 6.022 x10 23 atoms of co2?
Hence 6.022 X 1023 molecule of CO2 weighs 44 g.
What is the mass of 6.022 x10 23 atoms of oxygen?
of oxygen atoms = 1.5 × 6.022×1023 = 9.033×1023 atoms Mass of Oxygen atoms = 1.5 × 16 = 24 g.
How many moles are in 500 grams of iodine?
| Question | Answer |
|---|---|
| You discover that the head of a match contains 1.6 grams of Sulfur. How many atoms of Sulfur does a match contain? | 3.0 X 10^22 atoms S |
| While cleaning a cut, you spill a bottle of Iodine. The label says that the bottle holds 500 grams of I. How many moles of I are there? | 3.94 mol I |
How many atoms are in 32 grams of sulfur?
Answer and Explanation: 32 grams of sulfur will contain 6.022 X 1023 sulfur atoms.
How do you calculate atoms?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro’s number: 6.02 x 10^23.
Is mass number and atomic mass same?
The mass number is the sum of the number of protons and neutrons in an atom. It is a whole number. The atomic mass is the average number of protons and neutrons for all natural isotopes of an element. It is a decimal number.
What is in atomic mass?
Atomic mass indicates the size of an atom. Although technically the mass is the sum of the mass of all the protons, neutrons, and electrons in an atom, the mass of an electron is so much less than that of the other particles, that the mass is simply that of the nucleus (protons and neutrons).
What is the mass of sulfur-33?
sulfur-33 atom (CHEBI:37980) The stable isotope of sulfur with relative atomic mass 32.9714585, 0.75 atom percent natural abundance, and nuclear spin 3/2.
How To Convert Grams to Atoms – THE EASY WAY!
Images related to the topicHow To Convert Grams to Atoms – THE EASY WAY!

How many protons does sulfur 35?
Because it is S , the number of protons is 16 ; ordinarily you would not be given this info because you are expected to utilize the Periodic Table. If the isotopic mass is 35 , this sulfur isotope must contain 19 neutrons.
How many neutrons are in sulfur-32?
| Properties of Sulfur-32 Isotope: | SULFUR-32 |
|---|---|
| Neutron Number (N) | 16 |
| Atomic Number (Z) | 16 |
| Mass Number (A) | 32 |
| Nucleon Number (A) | 32 |
Related searches to what is the mass of exactly 500 sulfur atoms
- the internationally accepted atomic mass standard is carbon 12 because this isotope
- what is the mass of 4 moles of carbon mass is rounded to 1 decimal place
- what is the mass of exactly 500 sulfur atoms quizlet
- calculate the mass of 2 50 104 molecules of nitrogen gas
- what is the percent composition of na3po4
- calculate the mass percent of chlorine in ccl3f (freon-11).
- the internationally accepted atomic mass standard is carbon-12 because this isotope
- calculate the mass of 2.50 × 104 molecules of nitrogen gas.
- how many atoms are present in 0.375 mol of palladium (pd) atoms?
- how to find atomic mass of sulphur
- how many atoms are present in 0 375 mol of palladium pd atoms
- calculate the mass percent of chlorine in ccl3f freon 11
- what is the molar mass of carbon quizlet
- the mass of one mole of carbon dioxide is quizlet
Information related to the topic what is the mass of exactly 500 sulfur atoms
Here are the search results of the thread what is the mass of exactly 500 sulfur atoms from Bing. You can read more if you want.
You have just come across an article on the topic what is the mass of exactly 500 sulfur atoms. If you found this article useful, please share it. Thank you very much.