Are you looking for an answer to the topic “what is qh in thermodynamics“? We answer all your questions at the website barkmanoil.com in category: Newly updated financial and investment news for you. You will find the answer right below.
Qh is the heat transfer out of the hot reservoir, W is the work output, and Qc is the heat transfer into the cold reservoir.(QH = heat rejected to high temperature medium and QL = heat input from low temperature medium.)Heat (Qh) is absorbed from a source at a high T. Work is done by the engine. Heat (Qc) is expelled to a source at a lower T. Since it is a cyclic process, ∆U = 0. Thus, W = Qtotal —> W = Qh – Qc.

What does QH mean in thermodynamics?
(QH = heat rejected to high temperature medium and QL = heat input from low temperature medium.)
What is QH chemistry?
Heat (Qh) is absorbed from a source at a high T. Work is done by the engine. Heat (Qc) is expelled to a source at a lower T. Since it is a cyclic process, ∆U = 0. Thus, W = Qtotal —> W = Qh – Qc.
Physics – Thermodynamics: (1 of 14) Efficiency Of Heat Engines: Basics
Images related to the topicPhysics – Thermodynamics: (1 of 14) Efficiency Of Heat Engines: Basics

What is Q and Q in thermodynamics?
Heat in thermodynamics
Scientists define heat as thermal energy transferred between two systems at different temperatures that come in contact. Heat is written with the symbol q or Q, and it has units of Joules ( Jstart text, J, end text).
What is TH and TL in Carnot cycle?
A heat engine with Carnot cycle, also called Carnot heat engine, can be simplified by the following model: A reversible heat engine absorbs heat QH from the high-temperature reservoir at TH und releases heat QL to the low-temperature reservoir at TL. The temperatures TH and TL remain unchanged.
What is δu?
Macroscopically, we define the change in internal energy ΔU to be that given by the first law of thermodynamics: ΔU = Q− W. Many detailed experiments have verified that ΔU = Q − W, where ΔU is the change in total kinetic and potential energy of all atoms and molecules in a system.
What is QH energy?
We define Qh as. the heat pumped into cold reservoir, Qc heat taken from. (cold) outdoors, and W as the electrical energy used by. pump.
What is Q in Carnot cycle?
The Carnot cycle consists of the following four processes: A reversible isothermal gas expansion process. In this process, the ideal gas in the system absorbs qin amount heat from a heat source at a high temperature Thigh, expands and does work on surroundings.
See some more details on the topic what is qh in thermodynamics here:
What Is Qh In Thermodynamics – Fossil Hunters
The 1st Law for the cycle is W = QH – QL, where QH is the energy delivered to the gas by the nuclear reactor and QL is the heat rejected to the …
What does QH and QL actually mean : r/thermodynamics
QH is the heat energy rejected to the high temperature reservoir and likewise, QL is the amount of heat energy extracted from the low …
Thermodynamics/The Second Law of Thermodynamics
The thermodynamic temperature scale (absolute T)Edit. Efficiency of a Carnot engine is: η = 1 – (Qc / Qh) = 1 – (Tc / Th).
Chapter 5: The Second Law of Thermodynamics
Chapter 5: The Second Law of Thermodynamics. In this chapter we consider a more abstract approach to heat engine, refrigerator and heat pump …
What is CP value in thermodynamics?
In thermodynamics, the heat capacity ratio or ratio of specific heat capacities (Cp:Cv) is also known as the adiabatic index. It is the ratio of two specific heat capacities, Cp and Cv is given by: The Heat Capacity at Constant Pressure (Cp)/ Heat capacity at Constant Volume(Cv)
What does Q mean in chemistry?
What is Q? The reaction quotient Q is a measure of the relative amounts of products and reactants present in a reaction at a given time.
Why is Q used for heat?
An uppercase letter Q was first used to symbolize “the absolute quantity of heat” by the French engineer, Benoit-Paul-Émile Clapeyron (1799−1864), in his famous memoir of 1834 in which he first quantified what it is now known as the Carnot cycle (1).
What does Q stand for in Q MCT?
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K) ∆T = Change in temperature (Kelvins, K)
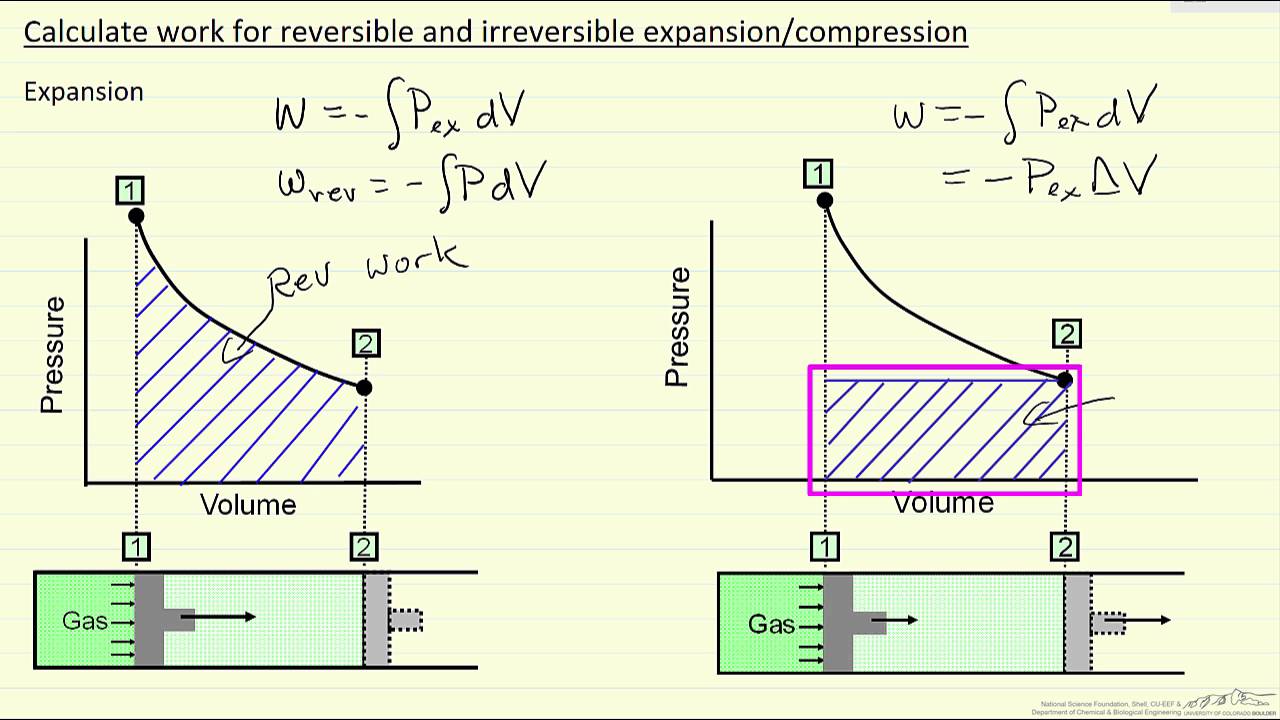
Calculate Work for Reversible and Irreversible Expansion/Compression
Images related to the topicCalculate Work for Reversible and Irreversible Expansion/Compression
What is Q in thermal conductivity?
The transfer of heat energy is defined as heat flux, Q. By definition, this is the flow of heat energy through a defined area over a defined time. So, the units for Q are Joules (energy) divided by area (square meters) and time (seconds).
How do you calculate TC in physics?
- Use the temperature converter to convert 25°C to Kelvins. You can also do the calculations manually; in this case, Tc = 25 + 273.15 = 298.15 K .
- Repeat step 1 for the temperature of the hot reservoir. …
- Input these values into the Carnot efficiency equation:
What is the formula of efficiency of Carnot engine?
efficiency =WQH=1−TCTH. These temperatures are of course in degrees Kelvin, so for example the efficiency of a Carnot engine having a hot reservoir of boiling water and a cold reservoir ice cold water will be 1−(273/373)=0.27, just over a quarter of the heat energy is transformed into useful work.
What is SI unit of entropy?
Entropy is a function of the state of a thermodynamic system. It is a size-extensive quantity, invariably denoted by S, with dimension energy divided by absolute temperature (SI unit: joule/K).
What is δu and ΔH?
ΔH is the thermodynamic potential of the system, and thus includes work done and ΔU, ΔH=ΔU + PΔV. ΔU is the summation of energies in a system. When work done=0, ΔH=ΔU.
When the first law of thermodynamics Q δu W is applied to an ideal gas that is taken through an isobaric process?
Applying the first law of thermodynamics, if ΔU=Q+W, and Q is 0, the change in internal energy of the gas must be equal to the work done on the gas (ΔU=W). In an isobaric process, pressure of the gas remains constant.
What is the formula for efficiency?
Efficiency can be expressed as a ratio by using the following formula: Output ÷ Input. Output, or work output, is the total amount of useful work completed without accounting for any waste and spoilage. You can also express efficiency as a percentage by multiplying the ratio by 100.
What does cop HP 1 represent?
In the spirit of taking the ratio of what you get to what you spend, we define a heat pump’s coefficient of performance (COPhp) to be COPhp=QhW C O P hp = Q h W . Since the efficiency of a heat engine is Eff=WQh E f f = W Q h , we see that COPhp=1Eff C O P hp = 1 E f f , an important and interesting fact.
What does the Q stand for in Q MC T?
Q = mc∆T. Here, Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K)
What is Q in Gibbs free energy?
Using Standard Change in Gibbs Free Energy, ΔG°
where R is the ideal gas constant 8.314 J/mol K, Q is the reaction quotient, and T is the temperature in Kelvin.
First Law of Thermodynamics, Basic Introduction – Internal Energy, Heat and Work – Chemistry
Images related to the topicFirst Law of Thermodynamics, Basic Introduction – Internal Energy, Heat and Work – Chemistry

What does Q stand for in calorimetry?
| Quantity | Symbol | Meaning |
|---|---|---|
| heat | q | Energy transfer that produces or results from a difference in temperature |
| temperature | T | Measure of the kinetic energy of molecular motiom |
| temperature change | DT | Difference between the final and initial temperatures for a process |
| mass | m | Amount of material present |
What does Q stand for in thermochemistry?
Apr 14, 2015. q=mcΔT , in which q is the energy gained or lost, m is mass, c is specific heat capacity, and ΔT is the change in temperature (final temperature – initial temperature).
Related searches to what is qh in thermodynamics
- qh formula
- what is qh and ql in thermodynamics
- second law of thermodynamics
- of the following which is not a statement of the second law of thermodynamics
- what is q thermodynamics
- what is th and tl in thermodynamics
- what is th and tc
- examples of 2nd law of thermodynamics
- what does q mean in thermodynamics
- Second law of thermodynamics
- what is q in thermodynamics
- cop thermodynamics
- eta thermodynamics
- what is qc and qh in thermodynamics
Information related to the topic what is qh in thermodynamics
Here are the search results of the thread what is qh in thermodynamics from Bing. You can read more if you want.
You have just come across an article on the topic what is qh in thermodynamics. If you found this article useful, please share it. Thank you very much.