Are you looking for an answer to the topic “smallest percent ionic character“? We answer all your questions at the website barkmanoil.com in category: Newly updated financial and investment news for you. You will find the answer right below.
Keep Reading

How do you find the lowest ionic character?
Therefore the smallest electronegativity difference (least ionic character) will be found between O-F, then C-F, then H-F, and finally the greatest electronegativity difference (most ionic character) will be between Na – F.
What is the percentage ionic character?
A bond’s percent ionic character is the amount of electron sharing between two atoms; limited electron sharing corresponds with a high percent ionic character. To determine a bond’s percent ionic character, the atoms’ electronegativities are used to predict the electron sharing between the atoms.
Example percent ionic character
Images related to the topicExample percent ionic character

How do you predict percent ionic character?
One method of estimating the percent ionic character is to set it equal to the ratio of the observed dipole moment to the value of eR, all multiplied by 100.
Which shows high percentage of ionic character?
Solution(By Examveda Team)
HF has the highest percentage of ionic character because F has the greatest electronegativity.
Which of the following has maximum ionic character LICL NaCl KCL CsCl?
According to Fajan’s rule: Bigger cation and smaller anion will favour ionic character. So, CsCl has maximum ionic character.
Which of the following covalent bonds has the highest percent ionic character?
Since electronegativity difference is highest between Cs and F,CsF has highest percentage of ionic character in its bonding.
Which of the following is least ionic?
Reason: AgCl having least covalent character and Ag having least ionic character.
See some more details on the topic smallest percent ionic character here:
CHAPTER 9 Flashcards | Quizlet
34) Identify the compound with the smallest percent ionic character. A) HF B) IBr. C) HCl. D) LiF. B.
Identify the compound with the smallest percent ionic… – Techwhiff
Answer: IBr has the smallest percentage ionic character amongst the given compounds.Explanation: Percentage ionic character is calculated by using the …
Smallest among these species is: – Toppr
33] The values of electronegativity of atoms A and B are 1.20 and 4.0 respectively. The percentage of ionic character of A – B bond is 1) 50 % 30. Doo 28 2) 43% …
Smallest among these species is: – Toppr
b omb 37. The values of electronegativity of atoms A and B are 1.20 and 4.0 respectively. The percentage of ionic character of A-B bond is 1) 50% 2) 72.24% 3) …
What is the percentage ionic character of NaCl?
As a result, NaCl’s ionic character percentage is 59.55 percent.
What is percentage ionic character from dipole moment?
Percentage ionic character = observed dipole momenttheoretical dipole moment×100=0.727.2×100=10%
How do you compare ionic characters?
The bigger difference between the electronegativity of two atom the ionic the character is. You can determine the ionic character by caunting the percent ionic character formula. , Tutor for chemistry. The ability to lose electrons (metals) or gain them (non-metals) and the behavior it exhibits while doing that.
How do you calculate percent ionic character using electronegativity?
One way of estimating the ionic character of a bond—that is, the magnitude of the charge separation in a polar covalent bond—is to calculate the difference in electronegativity between the two atoms: Δχ = χB − χA.
Which compound has the greatest degree of ionic character?
A bond between H and HCl will exhibited the greatest degree of ionic character. Cl is the most electronegative atom among the atoms present in the choices.
percent ionic character worked example problem
Images related to the topicpercent ionic character worked example problem

Which of the following has maximum covalent character LiI LIF LiCl LIBR?
Fajans rule states that the polarising power is directly proportional to the radius of anion. hence the bigger the anion the more covalent it is. Hence LiCl is the most ionic whereas LiI is most covalent.
Which of the following has highest ionic character MgCl2 CaCl2 bacl2 BeCl2?
Correct option b BeCl2 < MgCl2 < CaCl2 < BaCl2Explanation:Ionic character is decided by Fazans rule. More large size of cation more will be ionic character.
Which is more ionic LiCl or BeCl2?
BeCl2 is more covalent than LiCl. As per the Fajan’s rule, more the polarisation, more will be the covalent character.
Which of the following compounds has the most ionic bonding has the highest percentage of ionic character )?
Theoretically, the compound with the most ionic character is CsF (Cesium Fluoride), because it has the maximum ionic character. Fluorine being the most electronegative element with an electronegativity value of 4.0 (on the Pauling scale) and Cs being the least, form the bond with maximum ionic character which is 92%.
How do you find percent covalent character?
Example. Calculate the % of ionic character of a bond having length = 0.83 Å and 1.82 D as it’s observed dipole moment. The example given above is of a very familiar compound called HF. The % ionic character is nearly 43.25%, so the % covalent character is (100 – 43.25) = 56.75%.
What does ionic character depend on?
Ionic character depends upon, electronegativity difference between cation and anion of compound. As these difference increases, ionic character increases. Larger the size of cation and smaller the size of anion, more is the electronegativity difference and more is the ionic character.
Which of the following has lowest ionic character AgCl KCL bacl2 cacl2?
Here, $AgCl$ shows least ionic character. Elements have the tendency to gain and lose electrons to complete its octet and become stable and hence ions are formed.
What is ionic character?
Ionic character refers to the percentage of difference between the electronegativity of two covalently bonded atoms.
Which of the following is least ionic NaF NaCl NaBr NaI?
Polarizability (tendency of an anion to get distorted/polarized) of an anion increases with the increase in size of the anion. Thus, the Polarization and hence the Covalence increases with size of anion. Thus NaF is more ionic than NaI. NaF > NaCl > NaBr > NaI .
Which compound has the most ionic character?
- K-F would be the most ionic because K has an electronegativity of 0.82 and F has an electronegativity of 3.98.
- Ca-F would be next. …
- Br-F. …
- Cl-F.
Which of the following salts has least ionic character?
Answer: Magnesium chloride molecule is having least ionic character.
Dipole Moment, Molecular Polarity Percent Ionic Character
Images related to the topicDipole Moment, Molecular Polarity Percent Ionic Character
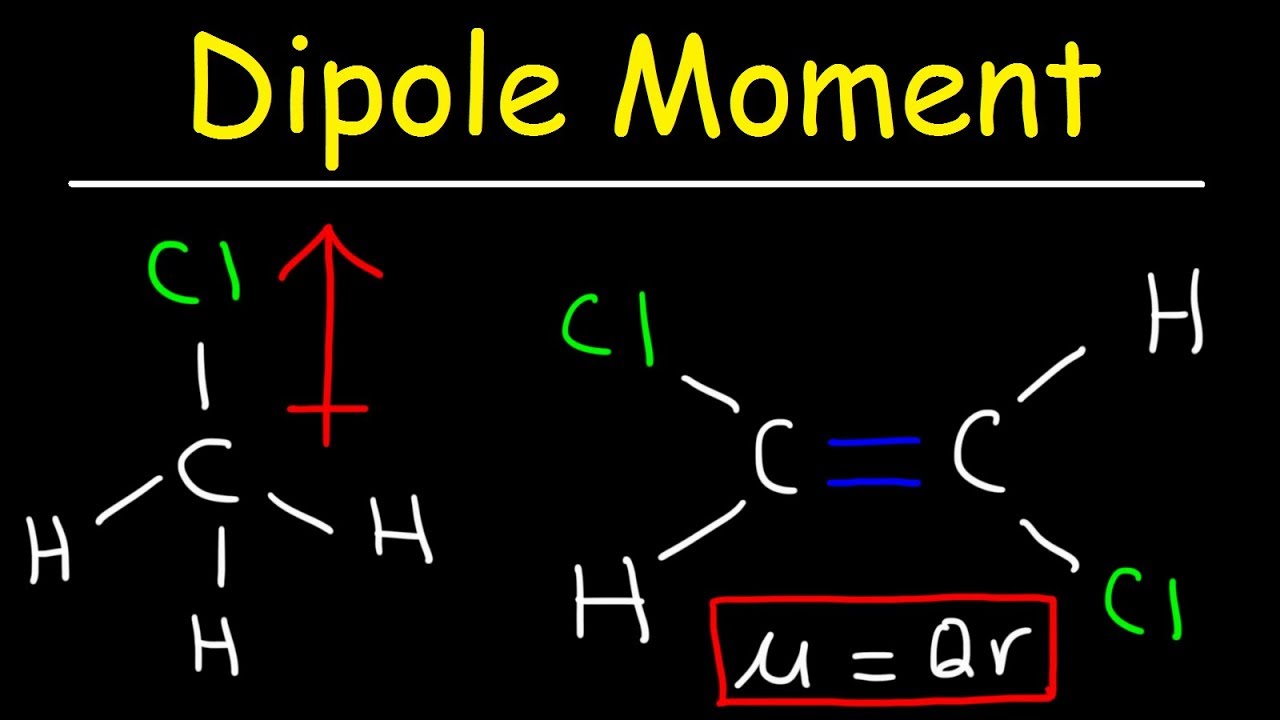
Which compound has the greatest degree of ionic character?
A bond between H and HCl will exhibited the greatest degree of ionic character. Cl is the most electronegative atom among the atoms present in the choices.
What is ionic character?
Ionic character refers to the percentage of difference between the electronegativity of two covalently bonded atoms.
Related searches to smallest percent ionic character
- identify the compound with the smallest percent ionic character
- percent ionic character calculator
- what does percent ionic character mean
- which of the following bonds is expected to have the smallest percent ionic character?
- choose the bond below that is least polar
- which of the following bonds is expected to have the smallest percent ionic character
- how many of the following elements can form compounds with an expanded octet i na br s
- what is the least ionic character
- electronegativity chart
- percent ionic character example
- which molecule or compound below contains an ionic bond?
- identify the compound with the highest percent ionic character
- which has lowest ionic character
- which of the following contains the most lattice energy a kcl b bacl2 c cas d na2o
- which molecule or compound below contains a pure covalent bond li2co3 scl6 cl2 pf3 nacl
- which molecule or compound below contains an ionic bond
Information related to the topic smallest percent ionic character
Here are the search results of the thread smallest percent ionic character from Bing. You can read more if you want.
You have just come across an article on the topic smallest percent ionic character. If you found this article useful, please share it. Thank you very much.